
Understanding the differences between GRAS and NDI is essential for any company developing food ingredients or dietary supplements in the U.S. market. Both pathways help you meet FDA safety requirements before launch. However, they apply to different product types and rely on distinct kinds of evidence.
Therefore, choosing the correct pathway early prevents delays, reduces costs, and strengthens your strategy. This guide explains what GRAS and NDI mean, how they differ, and how to decide which route fits your ingredient. Keep reading to learn how to stay compliant and move your product forward with confidence.
What Is GRAS?
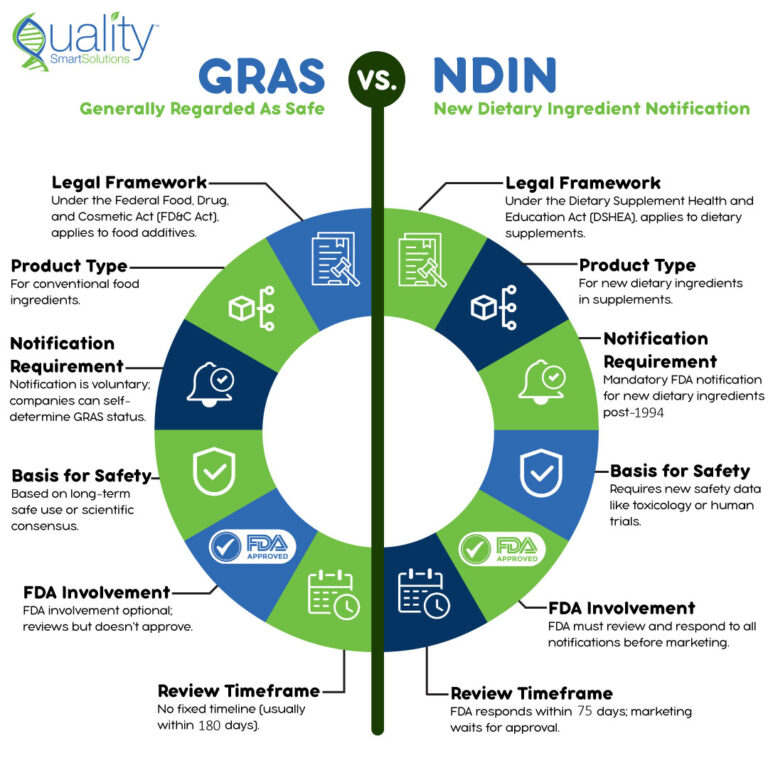
GRAS (Generally Recognized as Safe) covers substances added to conventional foods when qualified experts agree they are safe. Instead of relying on private files, you support the conclusion with publicly available data and a clear scientific rationale.
In practice, GRAS applies to ingredients used in conventional foods and follows the safety standard of a “reasonable certainty of no harm.” You can self-affirm GRAS or, alternatively, submit a GRAS notification to the FDA. If the FDA has no concerns, it issues a “no objection” response.
GRAS determinations typically draw on:
- Scientific procedures, including published toxicology, exposure, and clinical studies.
- Common use in food before 1958, which demonstrates long-term safe consumption.
For example, certain enzymes, plant extracts, and flavoring substances qualify when the public literature supports safety under the intended conditions of use.
What Is NDI?
NDI (New Dietary Ingredient) applies to ingredients used in dietary supplements, not conventional foods. Under the Dietary Supplement Health and Education Act (DSHEA), any dietary ingredient not marketed in the U.S. before October 15, 1994, generally requires a New Dietary Ingredient Notification (NDIN).
With NDIN, you notify the FDA at least 75 days before marketing and provide safety data for the specific use. Unlike GRAS, NDIN submissions may include proprietary or unpublished studies. Consequently, the pathway allows greater protection for trade secrets and formulation IP.
Additionally, NDI evaluations consider dose, target population, and use conditions. When data are limited, you may restrict certain groups, such as children or pregnant women, on the supplement label.
GRAS vs NDI: Key Differences
Although both pathways focus on safety, they serve different goals. The table below highlights the core distinctions to guide your choice.
Aspect | GRAS | NDI |
Applies to | Conventional foods | Dietary supplements |
Regulatory basis | Food Additives Amendment of 1958 | DSHEA, 1994 |
Notification to FDA | Voluntary | Mandatory |
Safety data | Must be publicly available | Can remain confidential |
Label restrictions | Cannot limit intake by population | Can exclude specific populations |
Use of ingredient | Allows use in food supply | Limited to supplement use |
Importantly, if an ingredient is GRAS for food use, you can often use the same ingredient in supplements without a new NDIN — provided the form, exposure, and conditions of use match the GRAS conclusion. Consequently, GRAS can create broader downstream flexibility.
Choosing the Right Pathway
To decide which route fits best, consider these practical questions:
1. Where will you use the ingredient?
a. Food or beverage: GRAS
b. Dietary supplement: NDI or GRAS (if already in the food supply)
2. What type of safety data do you have?
a. Publicly available data: GRAS
b. Confidential studies: NDI
3. Do you plan to restrict certain populations?
a. Yes: NDI
b. No: GRAS
4. Do you want FDA acknowledgment?
a. Optional under GRAS
b. Required under NDI
Therefore, if your ingredient meets GRAS criteria and enters the food supply, you may later use it in supplements without submitting an NDIN, as long as you stay within the established limits.
Key Takeaways
- GRAS vs NDI: differ mainly by product type (food vs. supplement.)
- GRAS relies on publicly available data, whereas NDIN can include confidential studies.
- FDA notification is mandatory for NDIN; for GRAS, it is optional via notification.
- GRAS can unlock use in both foods and supplements; NDIN is supplement-only.
- Choosing wisely early improves timelines, reduces risk, and supports long-term compliance.
Frequently Asked Questions
Is GRAS status easier to achieve than an NDIN?
Not necessarily. GRAS demands consensus from qualified experts and robust public data, which takes planning. That said, with the right evidence and a clear exposure assessment, you can reach a GRAS conclusion efficiently.
Can one ingredient be both GRAS and an NDI?
Sometimes brands assume the pathways are mutually exclusive. In reality, if an ingredient is GRAS for food use, you may also use it in supplements without an NDIN, provided the form and exposure match the GRAS conditions.
Do I need formal FDA approval for GRAS or NDIN?
No. Neither pathway requires approval. However, NDIN notification is mandatory for new dietary ingredients, and GRAS conclusions can be voluntarily submitted to FDA for added transparency.
What if I skip NDIN notification?
Skipping the required notification can trigger enforcement and market withdrawal. Fortunately, with early planning, you can choose the correct route and submit a strong package.
Next Steps
Ultimately, choosing between GRAS and NDI depends on your ingredient’s intended use, data availability, and target market. Both pathways play critical roles in ensuring product safety and consumer protection.
If you’re unsure which route fits your ingredient, our regulatory specialists can help. We provide complete support for GRAS submissions and NDI notifications to help you achieve compliance efficiently.
For personalized guidance, contact our team today and get expert help with FDA regulatory requirements.







