
Health Canada Fees 2026: Updates for Devices, Drugs & Cannabis
Health Canada has updated its fees effective April 1, 2026, with changes applying across multiple regulated sectors, including medical devices,
... Read more
Health Canada has updated its fees effective April 1, 2026, with changes applying across multiple regulated sectors, including medical devices,
... Read more
Arab Health 2025 is one of the world’s largest healthcare exhibitions. It brings together industry leaders, innovators, and regulatory experts
... Read more
Understanding the FDA classification system for medical devices is key to getting your product to market safely and efficiently. Knowing
... Read more
Are you trying to make sense of the FDA’s 510(k) pre-submission process? You’re not alone. Many companies, especially those new
... Read moreThe process of bringing medical devices to market in the United States involves stringent regulations imposed by the Food and
... Read more
https://youtu.be/E0E0dohMpC4 So, you’re a medical device manufacturer eyeing the Canadian market? Great choice! But before you dive in, let’s talk
... Read more
Introduction In the ever-changing world of medical device manufacturing, ensuring your product complies with regulations is akin to navigating stormy
... Read more
*This summary of Health Canada’s guidance document provides compliance details for manufacturers submitting a new or amended application. Under the
... Read more
Del Health Canada is a crucial regulatory body responsible for ensuring the safety and effectiveness of drugs and medical devices
... Read more
Quality Smart Solutions offers the best FDA Facility Renewal & Registration services in North America for Medical Devices, Food, and
... Read more
Navigating the complex medical device regulatory process can often feel like decoding a cryptic language. Manufacturers grapple with an intricate
... Read more
Are you a medical professional or someone involved in the healthcare industry? If so, you’ve likely come across the
... Read more
Introduction: FDA approvals can be a complex process, but understanding its important steps can give you insight into how drugs
... Read more
Introduction: Manufacturing and selling medical devices in Canada can be a complex process and requires adherence to Health Canada’s
... Read more
As a Canadian medical device license (MDL) holder, you are subject to an annual license renewal procedure. The renewal process
... Read more
The price for an MDEL (Medical Device Establishment Licence) and MDL (Medical Device Licence) in Canada varies according to the
... Read more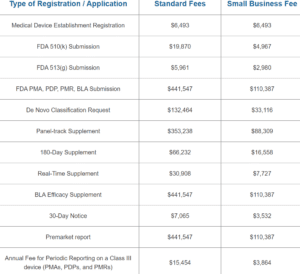
The cost to register an FDA Medical Device Establishment is USD $6,493 for 2023. The FDA’s fiscal year 2023
... Read more
Looking for a quality import service can be a daunting task, but with the right advice and tips, you can
... Read more
What are Class 1 Medical Devices? Class 1 medical devices are the lowest risk category of medical devices, as they
... Read more
Background: Health Canada issued three interim orders to allow the importation and sale of medical devices used to diagnose, treat,
... Read more
Introduction: Class 3 medical devices are subject to the highest level of scrutiny and require special attention when it comes
... Read more
Submitting a 510K Premarket Notification is an essential step in the process of getting medical devices approved by the
... Read more
Introduction: Medical devices are an important part of healthcare and play a critical role in the treatment of patients. Medical
... Read more
Navigating medical device regulations set by Health Canada can be challenging. However, knowing the correct classification of your medical device
... Read more
The Medical Device License is a legal document required to manufacture, sell, and distribute medical devices. The Medical Device Licence
... Read more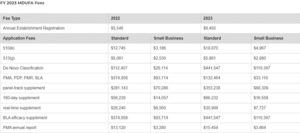
The FDA or U.S. Food and Drug Administration recently announced the Fiscal Year Medical Device User Fee (MDUFA) amendments. The
... Read more
The Medical Device License is a legal document that is required for the manufacture, sale, and distribution of medical
... Read more
Nowadays almost all the facets of healthcare are well-equipped with the modern and latest technology tools, including software systems.
... Read more
In response to the COVID-19 public health emergency, FDA issued a declaration regarding the appropriateness of utilizing emergency use authorizations
... Read more
On December 11, 2021, Health Canada issued a notice regarding the annual adjustment of fees for drugs and medical devices
... Read more
Interim Order No. 3 Respecting the Importation and Sale of Medical Devices for Use in Relation to COVID-19 (the third interim
... Read more
Regulatory agencies across the globe, including the U.S. Food and Drug Administration (FDA), Health Canada, and the United Kingdom’s Medicines
... Read more
Manufacturers and companies exporting medical devices from Canada may be asked by foreign customers or foreign governments to supply certification
... Read more
The advancement of technology is rapidly progressing, and the evolution can be observed all around us. As cutting-edge technology becomes
... Read more
It’s that time of year again! Any domestic and foreign establishments registered with US FDA must renew their registration annually
... Read more
On June 28, 2021, Health Canada opened a public consultation on a Unique Device Identification (UDI) system for medical devices
... Read more
Who Needs an MDSAP Certificate? Anyone looking to manufacture a Class II, III or IV medical device in Canada requires
... Read more
Health Canada has published a consultation paper on proposed changes to the regulation and oversight of clinical trials for drug,
... Read more
Cancellation of Medical Device Establishment Licence for A Non-compliance With Annual Licence Review Requirements On May 27, 2021 Health Canada
... Read more
As of March 1, 2021, Interim Order No. 2 replaces Interim Order No. 1 Respecting the importation and sale of
... Read more
Health Canada recently published an additional notice regarding all interim orders relating to COVID-19 and Medical Devices. This notice is
... Read more
Earlier this summer, Health Canada provided a notice to industry on the regulatory considerations for non-medical masks or face coverings,
... Read more
Health Canada is not only the national regulatory authority for issuing health product marketing authorizations, but it also oversees and
... Read more