The coronavirus-19 (COVID-19) pandemic has had a long-lasting effect on many of the processes done in business and everyday life. One of the resulting main effects is delays in application processing times. From simple applications such as passports to larger-scale applications such as NHP licensing.
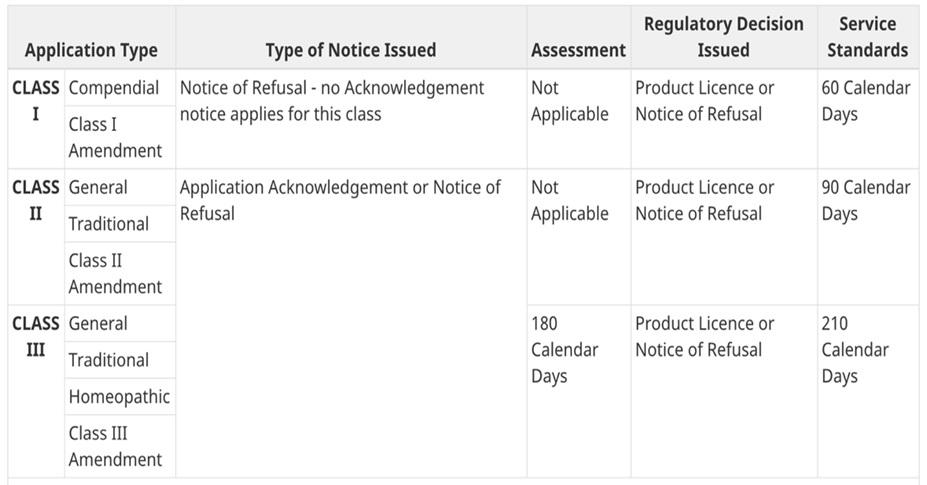
Prior to the pandemic, Natural and Non-Prescription Health Products Directorate (NNHPD) had a well-established service standard for Class I, II and III applications, which is outlined in the Management of Application Policy (MAP) guidance as shown below:
As the focus had started to shift to prioritize COVID-related product applications, a resultant de-prioritization of the non-COVID-related applications occurred. Not surprisingly, the volume of priority applications began to create a backlog on the non-priority product, site, and post-license submissions, thereby increasing the service standards. Within the past year, NNHPD has managed to achieve the following:
- NNHPD licensed 4,290 new alcohol-based hand sanitizers and issued 2,054 interim Covid site licenses.
- NNHPD issued 10,826 Non-COVID related product licence and 1,126 site licence decisions
To manage stakeholder expectations, NNHPD provided discretionary dates in which they hoped to see 90% of applications fulfill their original committed service standard. These dates were further broken down into application type and class. In addition, regular updates were provided by NNHPD via the Performance standards for NHP applications Stakeholder Meetings, with the most recent one being March 24, 2021.
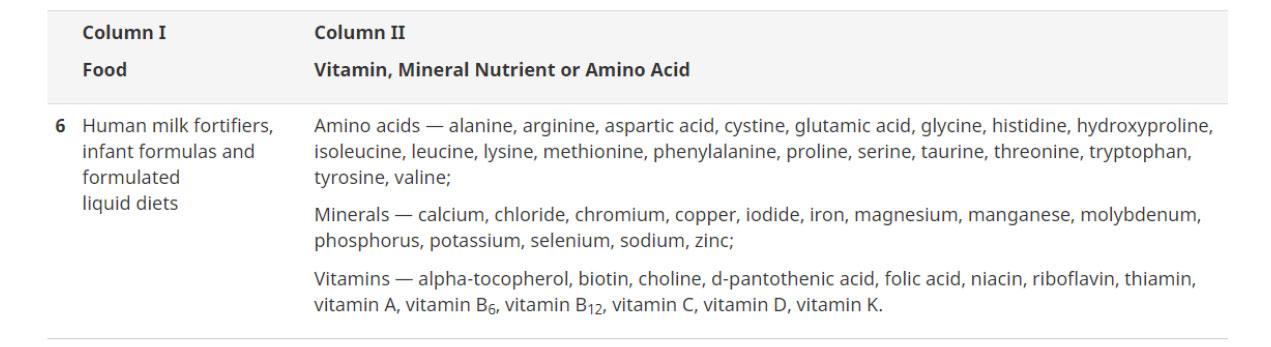
As of this fiscal year, NNHPD has already completed 5,046 Class I submissions, which is up 343 submissions from the previous year. The standard processing time for Class I submissions is 60 calendar days from the date of submission. NNHPD was able to meet the 90% target in months of January (93.8%), February (97.4 %), and March (91.4%).
For Class II, 2,221 submissions were completed in this fiscal year, which is up 301 submissions from the previous year. The standard processing time for Class II submissions is 90 calendar days from the date of submission. NNHPD was able to meet the 90% target in January (93.7%), February (93 %); however, they fell slightly below in March (89%).
Lastly, 3,298 Class III submissions were completed within the current fiscal year, which is up by 585 submissions from the previous year. Therefore, as a result, NNHPD is on track to meeting their 80% goal for the original 210 calendar day service standard, with over 88% of the backlog completed.
While much has been achieved, NNHPD has warned about the impacts of further volume increases will have on the current system. For example, a 40% increase in Class II and Class III applications was observed in March; thus, NNHPD has asked to limit submissions to critical applications.
Post-licensing submissions are subdivided into two categories: amendments and notifications.
Amendments are defined changes to an NHP that may have an impact on the safety, efficacy and/or quality of the product. In contrast, notifications are defined as changes to an NHP that do not significantly impact the product’s safety, efficacy, and/or quality. Similar to new product submissions, amendments follow the 3 Class application system and service standard. On the other hand, notifications have no established service standard and do not follow the 3-class system.
Only 63.9% of the received amendments have been issued a decision. There are approximately 147 submissions received in 2020, which remain to be processed. Similarly, 62% of received notifications have been completed to date.
The target has been successfully met for both Stream I (94%) and Stream II (96%) site license applications. Approximately 201 Stream I submissions, 389 Stream II submissions, and 318 renewals have been completed this year. In addition, extensions were granted for renewals to accommodate further processing.
Although it may seem that little can be done as a stakeholder to speed up the licensing process, there is one way, using the most up-to-date licensing forms. Within the last year, NNHPD has launched online webforms to be used in application submission. The web-based Product Licence Application (web PLA) and the amendment and notification form (ANF). The forms have a validation component built-in, which thereby lessens the burden for NNPHD. A lower burden allows for greater processing times and avoids unwarranted application refusals.
The ever-present changes resulting from COVID make it increasingly challenging to keep up with the most up-to regulatory information. As planning is a large part of the product launch, it is crucial to be in the know on impacts to your product plan. Our Experts at Quality Smart Solutions can offer support to your needs for foods, cosmetics, OTC drugs and medical devices for North America.
Contact us today to learn more about how we can support your compliance needs during and after licensing!