
GMP, GPP and GACP Standards That Strengthen Your Cannabis Business
If you are preparing a new cannabis product for market or expanding into a new jurisdiction, understanding GMP, GPP, and GACP
... Read more
If you are preparing a new cannabis product for market or expanding into a new jurisdiction, understanding GMP, GPP, and GACP
... Read more
If you manage regulatory submissions for natural health products, you likely already know that Health Canada ends FSRN acceptance as of February 16, 2026. Health Canada announced this change through Natural Health Products Site Licensing and Good Manufacturing Practices Inspection Bulletin No. 10.
... Read more
Health Canada is moving to answer a question the industry has been pressing for years: why should Canada conduct a
... Read more
Missing your MDEL renewal deadline puts your entire Canadian market access at risk. Under the Medical Devices Regulations (SOR/98-282), all Medical Device
... Read more
Health Canada has confirmed that Sports Electrolyte Supplements will be reclassified under a single regulatory framework, marking a significant change for products
... Read more
In December 2025, Health Canada finalized a major set of regulatory updates affecting controlled substances, cannabis, precursor chemicals, and certain drug manufacturing equipment.
... Read more
Health Canada has lowered the permitted level of caffeine and caffeine citrate in cola-type beverages from 200 ppm to 150
... Read more
Health Canada released a new modification to the List of Permitted Supplemental Ingredients that expands when and how caffeine can be
... Read more
NNHPD has introduced two new regulatory tools designed to simplify the natural health product submission process and give you more
... Read more
Health Canada has launched a new national consultation that could reshape how infant foods for special dietary purposes are assessed
... Read more
Health Canada has officially postponed the implementation of its proposed NHP cost recovery fees, which were initially expected to take
... Read more
When it comes to importing human food and animal food into Canada, many businesses assume the process is similar. In
... Read more
Getting an NPN for sports supplements is essential for businesses looking to market their products legally in Canada. The Natural
... Read more
When exporting food supplements to Canada, understanding trade agreements can make a huge difference in your profitability. The Comprehensive Economic
... Read more
On September 4, 2025, Health Canada released Version 4.0 of the Good Manufacturing Practices (GMP) Guide for Natural Health Products
... Read more
Businesses in Canada have long raised concerns about red tape in Health Canada regulations, from outdated rules to lengthy approval
... Read more
What is GMP training, and why does it matter? If your company makes, packages, labels, or imports health products, this
... Read more
Planning to sell a medical device under your brand in Canada? You might assume that using a licensed manufacturer means
... Read more
Over the past few weeks, many natural health product (NHP) companies have been caught off guard by an increase in
... Read more
Introduction Sodium reduction in processed foods is no longer optional. It is a critical step for industry players to safeguard
... Read more
When it comes to audits related to Health Canada requirements, staying compliant is about more than checking off a list.
... Read more
If you are planning to market animal health products, the question of pet supplements or veterinary health products will come
... Read more
Starting June 9, 2025, Class II NHP submissions will no longer be reviewed just based on the date you filed
... Read more
Thinking of selling hydration products in Canada? If your product makes a therapeutic claim, you may need a Natural Product
... Read more
Understanding the distinction between traditional cosmetics vs. cosmeceuticals is essential if you’re manufacturing or marketing skincare or beauty products in
... Read more
If you’re planning to sell or distribute a medical device in Canada, you’re going to hear two terms over and
... Read more
If your team handles drug product submissions in Canada, there’s a recent update from Health Canada you might want
... Read more
The eczema product market continues to grow as consumer demand increases, but that doesn’t mean you can jump in without
... Read more
How FDA and Health Canada Regulate Cosmetics Differently Thinking about launching your cosmetic line in North America? Whether you’re a
... Read more
Medical device shortage reporting requirements have recently changed, and it is essential for manufacturers and importers to stay informed. On
... Read more
Food additive submission may not be the flashiest part of launching a new food product in Canada, but it’s one
... Read more
Selling cosmetics on Amazon Canada can open up a world of opportunity, but it comes with strict compliance expectations. Recently,
... Read more
If you’re planning to launch a product like a protein bar, electrolyte drink, or mushroom supplement in Canada, it’s important
... Read more
If you plan to sell a natural health product in Canada, you’ve probably asked what an NPN is and
... Read more
If you’re trying to bring a food or supplement product to both the Canadian and US markets, you’ve probably already
... Read more
Health Canada has released a draft guidance document on co-packaged drug products, now open for public consultation. The guidance explains
... Read more
Introduction Recently, Health Canada announced interim policies affecting fresh coconut products and certain ready-to-eat breakfast cereals. The FOP labelling updates
... Read more
Changes are on the horizon for caffeine as a supplemental ingredient, and they could directly impact your business. Whether you
... Read more
Health Canada is conducting a public consultation on CBD regulations for natural health products, exploring a pathway to allow non-prescription
... Read more
The Natural and Non-prescription Health Products Directorate (NNHPD) has changed the review process for Class III Natural Health Product (NHP)
... Read more
Are Your Supplemental Ingredients Compliant? Canada’s New Rules Explained If your business sells supplemented foods in Canada, major regulatory updates
... Read more
The regulatory landscape for NHP labelling is evolving, with businesses facing increasing pressure to comply with Health Canada’s updated regulations. Initially
... Read more
Introduction Health Canada’s new front-of-package label rules are set to take effect on January 1, 2026, marking a significant shift
... Read more
Health Canada has made an important decision regarding grape seed extract, now approved for use as a supplemental ingredient in
... Read more
Navigating Health Canada’s rules for probiotic health claims can feel overwhelming, especially with the growing demand for these products. Businesses
... Read more
Learn about Health Canada’s transition strategy for infant formula regulations and ensure compliance before the December 31, 2024, deadline.
... Read more
Are you looking to bring your infant formula to the Canadian market? Successfully navigating Canada infant formula registration is essential
... Read more
If you’re thinking about bringing a pharmaceutical product to the Canadian market, getting familiar with drug submission pathways is a
... Read more
Health Canada has just released a draft Good Manufacturing Practices (GMP) guide for natural health products (NHPs), marking a monumental
... Read more
Designing a compliant food label is one of the most important steps when entering the Canadian market. The food labeling
... Read more
This fall, Health Canada is anticipating sharing a policy consultation document about health products containing (CBD). This proposal has been
... Read more
https://youtu.be/QABD-2XIRz0 Canada is known for its diverse culinary landscape. If you’re considering selling food products in this country, you’re in
... Read more
Introduction: As a food manufacturer in Canada, ensuring that your product labels comply with Health Canada’s regulations is crucial for
... Read more
Health Canada Approves Alpha GPC in Foods On September 20, 2023, Health Canada approved the use of L-alpha-glycerylphosphorylcholine (alpha GPC)
... Read more
From FDA guidelines to global regulatory standards, understanding the nuances of each requirement is essential to ensure a timely
... Read more
Using a medical device import agent can offer several benefits, especially if you are navigating the complex process of
... Read more
Introduction Health Canada MDALL is the key to legally marketing medical devices in Canada. If you are a manufacturer looking
... Read more
If you’re looking to grow your e-commerce business, learning how to sell on Amazon Canada is a smart move. With
... Read more
Introduction: Welcome to the ultimate guide to conducting a successful clinical trial, where we will delve into the best practices
... Read more
In today’s competitive marketplace, selling products on Amazon has become a lucrative opportunity for many entrepreneurs and businesses. However, navigating
... Read more
Introduction Navigating Health Canada’s licensing requirements can feel overwhelming, especially when your business depends on compliance to operate. Many companies struggle to
... Read more
In the ever-evolving healthcare landscape, staying up-to-date with the latest regulations and requirements is crucial for health professionals. And when
... Read more
Consumers rely on Health Canada monographs to ensure the safety and efficacy of their products. But have you ever wondered
... Read more
Introduction: When it comes to the pharmaceutical industry, there is no room for error. The production of pharmaceuticals requires strict
... Read more
Learn how to get your Medical Device Establishment Licence faster and stay compliant with Health Canada.
... Read more
Introduction: By extending the interim policy on the importation and sale of infant formulas, human milk fortifiers, and dietary products
... Read more
If you’re a business owner looking to expand your reach into the Canadian market, importing products may be a viable
... Read more
Health Canada has the authority to set and charge health product fees under the Food and Drugs Act (FDA). On
... Read more
https://youtu.be/dVbug89UHBA HACCP (Hazard Analysis and Critical Control Points) certification is a food safety management system that helps businesses identify and
... Read more
https://youtu.be/dVbug89UHBA In the food industry, ensuring the safety of the products you produce is of utmost importance. One key aspect
... Read more
As technology continues to advance, the use of Software as Medical Devices (SaMD) is becoming more prevalent in the healthcare
... Read more
https://youtu.be/mWaBQzsa1_M Introduction Importing goods can be a complex process, especially when it comes to navigating customs regulations and compliance requirements.
... Read more
Introduction: Manufacturing and selling medical devices in Canada can be a complex process and requires adherence to Health Canada’s
... Read more
As a Canadian medical device license (MDL) holder, you are subject to an annual license renewal procedure. The renewal process
... Read more
If you plan to manufacture, import, or sell a natural health product (NHP) in Canada, you need to understand the
... Read more
Looking for a quality import service can be a daunting task, but with the right advice and tips, you can
... Read more
PMRA Annual Fees reduced for Reporting and Applications: Health Canada is implementing a new approach beginning April 1, 2023. The
... Read more
Introduction: The sections relating to the disclosure of fragrance allergens because of the regulations go into effect two years following
... Read more
Introduction: Submitting a new drug to Health Canada can be a complex and time-consuming process. This post will provide
... Read more
Introduction Health Canada oversees the importation and sale of Veterinary health products (VHPs) in Canada through the VHP Notification Program.
... Read more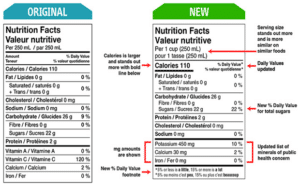
The Canadian Food Inspection Agency (CFIA) protects Canadians’ health and safety by ensuring food that reaches customers is safe, nutritious, and
... Read more
Top Canada Cosmetic Regulations Cosmetic Notification Form (CNF) Canada is one of the most popular tourist destinations in the world,
... Read more
Health Canada Announcement: The Minister of Health has introduced new front-of-package nutrition labeling laws for prepackaged food products, and
... Read more
The Canadian government has a set of regulations in place for natural health products (NHPs). These regulations help to ensure
... Read more
If you’re selling natural health products in Canada, understanding NHPID and NPN is essential. These tools from Health Canada determine
... Read more
Overview Manufacturers of prescription and non-prescription drugs are required to obtain a Drug Identification Number (DIN) before the drug product
... Read more
Who needs a Foreign Site Reference Number (FSRN)? For Natural Health Products (NHPs) manufactured, packaged, and/or labeled by sites located
... Read more
Learning how to classify your product correctly is one of the most important first steps when entering the Canadian market.
... Read more
Background On April 22, 2022, Health Canada issued a notice to the industry on the interim implementation of electronic
... Read more
What is a cosmetic? Under Section 2 of the Food and Drugs Act, a cosmetic is defined as “any substance
... Read more
Introduction: The market for animal and pet products is growing at a rapid pace. With that, there is also a
... Read more
Filling out a Cosmetic Notification Form is a required step before selling any cosmetic product in Canada. Health Canada uses
... Read more
On January 19, 2022, Health Canada announced a marketing authorization (MA) to permit manufacturers to voluntarily increase the amount of
... Read more
Regulatory agencies across the globe, including the U.S. Food and Drug Administration (FDA), Health Canada, and the United Kingdom’s Medicines
... Read more
Manufacturers and companies exporting medical devices from Canada may be asked by foreign customers or foreign governments to supply certification
... Read more
What is a Cosmetic? Cosmetics are a part of just about everyone’s life. Every day, most of us use products
... Read more
Pet Foods and Human Food Labels are designed to look similar yet distinct. There are some similar label requirements and
... Read more
Inspection of your food facility will help identify and fix any quality or processing deviations found regarding food safety. Review
... Read more
The “Sunshine” vitamin, Vitamin D, Vitamin D3 or Vitamin D2 as it is more commonly known. The term “sunshine” was
... Read more
What is a Cosmetic? Under Section 2 of the Food and Drugs Act, a cosmetic is defined as “any substance
... Read more