FDA Updates Yogurt Standard: Key Changes for Manufacturers
Attention all yogurt makers, big and small! The U.S. Food and Drug Administration (FDA) has finalized changes to the standard
... Read moreAttention all yogurt makers, big and small! The U.S. Food and Drug Administration (FDA) has finalized changes to the standard
... Read more
When it comes to introducing new substances into the world of food and dietary supplements, ensuring safety is paramount. The
... Read moreThe process of bringing medical devices to market in the United States involves stringent regulations imposed by the Food and
... Read more
https://youtu.be/dVbug89UHBA Introduction Ensuring food safety is crucial for businesses in the food industry. That’s where Hazard Analysis and Critical Control
... Read more
If you’re introducing a new ingredient or additive into food products, understanding the GRAS Notice vs. Self-Affirmed GRAS pathways is
... Read more
GRAS substances, or “Generally Recognized as Safe” substances, are essential to product development in the food, pharmaceutical, and cosmetic industries.
... Read more
Introducing the Supplemented Foods Facts Table (SFFT), your go-to guide for navigating the ever-changing world of supplemented foods. With consumer
... Read more
https://youtu.be/E0E0dohMpC4 So, you’re a medical device manufacturer eyeing the Canadian market? Great choice! But before you dive in, let’s talk
... Read more
https://youtu.be/QABD-2XIRz0 Canada is known for its diverse culinary landscape. If you’re considering selling food products in this country, you’re in
... Read more
Navigating the intricate world of medical devices can be daunting, especially when it comes to ensuring their safety and effectiveness.
... Read more
Drug registration and NDC number might sound technical, but they play a big role in how medications make it to
... Read more
Introduction In the ever-changing world of medical device manufacturing, ensuring your product complies with regulations is akin to navigating stormy
... Read more
Introduction In the world of medical devices, ensuring patient safety and efficacy is paramount. The 510(k) submission process is a
... Read more
Introduction: As a food manufacturer in Canada, ensuring that your product labels comply with Health Canada’s regulations is crucial for
... Read more
As a business owner operating in dietary supplements and health products, you are undoubtedly aware of the ever-evolving regulatory
... Read more
https://youtu.be/MlufAYFqQ1s In dietary supplements, navigating the intricate regulatory landscape is a challenging yet indispensable aspect of launching your product. Submitting
... Read more
https://youtu.be/x4c0LEPmJuM If you’re selling food products in the United States, understanding and complying with FDA regulations is non-negotiable. The Food
... Read more
When it comes to selling natural health products (NHPs) in Canada, securing an NPN license is your ticket to success.
... Read more
Introduction: Staying up-to-date with the latest regulations and tools is crucial in medical device submissions. As of June 9, 2023,
... Read more
The dietary supplement industry in the U.S. is growing fast. More people are looking for vitamins, minerals, and other supplements
... Read more
For importers in the global food trade, ensuring the safety and compliance of imported products is a top priority. The
... Read more
*This summary of Health Canada’s guidance document provides compliance details for manufacturers submitting a new or amended application. Under the
... Read more
The Food Safety Modernization Act (FSMA) Made Simple Imagine a world where preventing food-related illnesses is more important than dealing
... Read more
https://youtu.be/T3pLp1dF08Y Introduction: In the intricate world of global trade, ensuring the safety and compliance of imported and exported food products
... Read more
From FDA guidelines to global regulatory standards, understanding the nuances of each requirement is essential to ensure a timely
... Read more
In the realm of food production and processing, safety is paramount. Ensuring our food is free from contaminants and meets
... Read more
Introduction: So you’ve got a game-changing medical device that could reshape healthcare as we know it. Amazing! But before
... Read more
https://youtu.be/dVbug89UHBA In today’s fast-paced and diverse industries, pursuing safety and quality isn’t just a goal; it’s a non-negotiable commitment. This
... Read more
Introduction: Navigating international trade regulations and the intricacies of Importer of Record (IOR) compliance and taxes can be complex. This
... Read more
Understanding Food Labels: A Simple Guide Food labels are more than just ingredient stickers on packaging; they are a key
... Read more
Del Health Canada is a crucial regulatory body responsible for ensuring the safety and effectiveness of drugs and medical devices
... Read more
Health Canada, the Canadian federal health department, oversees the regulation of controlled substances in Canada. Controlled substances are regulated due
... Read more
Quality Management Systems (QMS) are an essential part of the life sciences healthcare industry, and many people need clarification on
... Read more
Are you ready to take your business to new heights? Look no further. In this comprehensive guide, we will unlock
... Read more
Health Canada publishes product monographs to describe a product’s health risks, benefits, and performance. Product monographs are for health products
... Read more
Using a medical device import agent can offer several benefits, especially if you are navigating the complex process of
... Read more
Quality Smart Solutions offers the best FDA Facility Renewal & Registration services in North America for Medical Devices, Food, and
... Read more
Introduction: The European Food Safety Authority (EFSA) provides regulatory guidance and oversees the approval process for novel foods in the
... Read more
Navigating the complex medical device regulatory process can often feel like decoding a cryptic language. Manufacturers grapple with an intricate
... Read more
Food manufacturers everywhere are willing to jump through hurdles to receive their GRAS Notifications and access to the generally recognized
... Read more
https://youtu.be/uuXA8yHrrZc Introduction: Behold the “Modernization of Cosmetic Regulation Act of 2022” that President Biden signed on December 29, 2022. This
... Read more
Are you looking to bring your natural health product to market? Navigating the process of obtaining a license can be
... Read more
In today’s rapidly evolving regulatory landscape, businesses face many challenges regarding ensuring compliance and product safety. One crucial aspect often
... Read more
Introduction Health Canada MDALL is the key to legally marketing medical devices in Canada. If you are a manufacturer looking
... Read more
Are you a health and wellness entrepreneur looking to bring your products to the Canadian market? Navigating the Health Canada
... Read more
Opening a drug testing facility can be a rewarding opportunity especially as demand for workplace and healthcare-related testing continues to
... Read more
If you’re looking to grow your e-commerce business, learning how to sell on Amazon Canada is a smart move. With
... Read more
In the dynamic and ever-evolving world of pharmaceuticals, ensuring compliance with FDA regulations is paramount. One crucial aspect that must
... Read more
Are you a medical professional or someone involved in the healthcare industry? If so, you’ve likely come across the
... Read more
In a world where culinary boundaries are constantly expanding, the future of food holds endless possibilities. Enter EFSA Novel Foods,
... Read more
In today’s global economy, businesses increasingly use international trade to source materials and sell products. However, the complex regulations
... Read more
Introduction: Quality assurance is an essential part of clinical trials. It involves a set of processes that ensure that the
... Read more
As a researcher, you know that the success of your study depends on many factors, from the quality of your
... Read more
As the saying goes, prevention is always better than a cure. This is particularly true regarding food safety in the
... Read more
As a company developing new products, ensuring they are safe for consumption is essential. One way to achieve this is
... Read more
Food safety is a top priority for Canadians. With so many potential hazards lurking in our food supply, it’s more
... Read more
Consumers today are more careful about the food they buy.They want to know their food is safe, high quality, and
... Read more
Introduction In the world of international trade, importers of record play a crucial role in ensuring smooth customs clearance and
... Read more
https://youtu.be/cct8v4aXvhc In the fast-paced and ever-evolving world of pharmaceuticals, staying ahead of the competition is crucial. And one vital
... Read more
https://youtu.be/dVbug89UHBA Welcome to a comprehensive Hazard Analysis and Critical Control Points HACCP process exploration. In today’s world, ensuring the safety
... Read more
Non-Conformance Reports Introduction: In today’s fast-paced and competitive business landscape, identifying and resolving issues is crucial for maintaining the
... Read more
Introduction to Cosmetic Regulations by the FDA: Are you a cosmetics brand looking to navigate the complex world of FDA
... Read more
When thinking about NDI Classification have you ever heard about the New Dietary Ingredient Notification in Ingredient Compliance? In today’s
... Read more
Are you a business owner in the health and wellness industry looking to navigate the complex regulatory landscape in Canada?
... Read more
In marketing, the power of persuasive structure-function claims cannot be underestimated. Regarding consumer perception and product sales, structure-function claims have
... Read more
In a world where the pharmaceutical industry is constantly evolving and new drugs are being introduced to the market every
... Read more
Are you a food, beverage, drug, or cosmetics business owner?If so, you already know how crucial it is to comply
... Read more
Introduction: Welcome to the ultimate guide to conducting a successful clinical trial, where we will delve into the best practices
... Read more
In today’s competitive marketplace, selling products on Amazon has become a lucrative opportunity for many entrepreneurs and businesses. However, navigating
... Read more
If you’re in the food business, you’ve likely come across the term “GRAS”, short for “Generally Recognized as Safe.” It’s
... Read more
Introduction Navigating Health Canada’s licensing requirements can feel overwhelming, especially when your business depends on compliance to operate. Many companies struggle to
... Read more
Are you a company in the healthcare or pharmaceutical industry looking to bring a new product to market? If so,
... Read more
In the ever-evolving healthcare landscape, staying up-to-date with the latest regulations and requirements is crucial for health professionals. And when
... Read more
https://youtu.be/mWaBQzsa1_M Introduction Welcome to a fascinating journey through the world of imported foods in Canada, where flavors from all
... Read more
Are you a business owner in the health and wellness industry? If so, you’re likely familiar with Health Canada’s Natural
... Read more
Proper documentation is essential in any business operation and is particularly crucial when it comes to PMA submissions. Preparing and
... Read moreThe healthcare industry is constantly evolving, and with the introduction of new technologies, it is becoming more efficient and effective
... Read more
Consumers rely on Health Canada monographs to ensure the safety and efficacy of their products. But have you ever wondered
... Read more
Introduction: When it comes to the pharmaceutical industry, there is no room for error. The production of pharmaceuticals requires strict
... Read more
As a business owner or operator, you may know the importance of ensuring that your products and services meet regulatory
... Read more
As more and more people become health-conscious, the demand for supplements has skyrocketed. One of the most popular supplements on
... Read more
Medical devices play a crucial role in modern healthcare, providing patients with life-changing treatments and improving the quality of
... Read more
As a business owner in the medical industry, you understand the importance of providing quality products and services to your
... Read more
Learn how to get your Medical Device Establishment Licence faster and stay compliant with Health Canada.
... Read more
Are you running a business that deals with food, drugs, medical devices, cosmetics, or dietary supplements? If so, you must
... Read more
In today’s global food supply chain, ensuring the safety and quality of food products is critical to protecting public health
... Read more
The healthcare industry is constantly evolving, and one of the most significant advances has been implementing drug identification numbers (DINs).
... Read more
Importing goods from other countries can be a lucrative business and a complex and tedious process. As an importer, you
... Read more
As a medical device manufacturer, getting your product to market can be a complex process with various regulatory requirements to
... Read more
If you’re a business owner looking to expand your reach into the Canadian market, importing products may be a viable
... Read more
In the pharmaceutical industry, proper drug submission management is crucial to the success of any drug development process. The submission
... Read more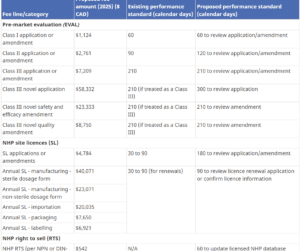
Health Canada has the authority to set and charge health product fees under the Food and Drugs Act (FDA). On
... Read more
The food industry has undergone a significant transformation in terms of safety and quality control over the years. From the
... Read more
Are you considering getting your food safety certification? In today’s highly competitive food industry, ensuring your food products are safe
... Read more
https://youtu.be/dVbug89UHBA HACCP (Hazard Analysis and Critical Control Points) certification is a food safety management system that helps businesses identify and
... Read more
As our world becomes increasingly globalized, the importation of food has become a common practice. However, with growing concerns
... Read more
Introduction: In the dynamic realm of food innovation, ushering in new ingredients responsibly and securely is of paramount importance. This
... Read more
https://youtu.be/yvnTszySkgg If you’re in the medical device manufacturing business, you’re likely familiar with the FDA’s regulations, including FDA Furls. However,
... Read more
https://youtu.be/iVJlCZmDeWU Introduction: If you’re a food manufacturer, you need to understand the generally recognized as safe (GRAS) process. This process
... Read more
https://youtu.be/NN8ZLynq6kE If you work in the food service industry, you may need to obtain food handlers license to ensure that
... Read more